About Our Patent
The United States Patent and Trademark Office (USPTO) has issued Bill Gardner a patent for the use of colored glass filters in UV LED Flashlights. My US Patent #7781751 Titled "Portable Wavelength Transforming Converter For UV LEDs" issued in August of 2010. The patent specifically defines band pass filters as being wavelength transforming materials. Here is a link: https://patents.google.com/patent/US7781751.
Excerpted from US Pat. #7781751
"The WT materials (14-14 n below) of any preferred embodiments may comprise any material or system that absorbs light of one wavelength or band of wavelengths and emits light of another wavelength or band of wavelengths, thus modifying the distribution of spectral density. Such materials or systems include band pass filters, phosphors and include also such materials or systems such as quantum dots whose properties are a combination of bulk properties and their physical particle size."The colored glass filter clearly absorbs light of one wavelength or band of wavelengths and emits light of another wavelength or band of wavelengths which is why a filter is used in the first place. It is obvious that the colored glass filter absorbs the visible light. It is also obvious that it is a different band of wavelengths that is emitted. It is less obvious, but the visible light that the filter absorbs does not just vanish, it is transformed into heat/infrared and re-emitted as infrared radiation. So not only is the band of wavelengths different on the emitted side of the colored glass filter, but also the visible radiation is transformed into infrared radiation.
History of the Invention
In 2006, I had a customer who asked about the buying UV LED flashlights. A source was found for some UV LED flashlights purporting to have a peak wavelength of 385 nm (with a maximum of 390 nm and a minimum of 380 nm). I saw that the UV LEDs were also emitting fairly large quantities of visible light. I had colored glass band pass, (visible light blocking UV transmitting), filter material available because I have been manufacturing Way Too Cool UV lamp fixtures since 1998. My company, Way Too Cool, immediately started selling UV LED flashlights with colored glass band pass filters installed starting in 2006. I was the very first person or company to manufacture and sell filtered UV LED Flashlights otherwise I would not have been able to get a patent on the innovation.
During the recent lawsuit, prior art was claimed trying to invalidate my patent and it was claimed that the filters were not wavelength transforming. In fact, even my own previous patents were considered prior art. Out of the many patents that the patent examiner researched, there were several patents that he did not examine, and these were put forth trying to show that my patent should be invalidated. However, the content of these additional patents was very similar to the patents that were examined and none of those patents teach the same things that my patent does. None of the prior art was conclusive at invalidating my patent. Further, they could not dispute that the colored bandpass filters were in fact wavelength transforming by converting the absorbed energy into IR photons.
Are absorption filters wavelength transforming?
During the recent lawsuit, neither of the Engenious Designs "Expert Witnesses" explained what happens to the energy that gets absorbed in the colored glass filters. The engineer from Hoya Optics also has no idea what happens to the energy that is absorbed.
However, my "Expert Witness" AND the textbooks on infrared astronomy explain that the visible light and UV radiation that the filter absorbs does not just vanish, but some of it is transformed/converted and re-emitted as photons of infrared radiation and some of it heats up the material. So not only is the band of wavelengths different on the emitted side of the colored glass filter, but also the visible light and some of the UV radiation is converted/transformed into photons of infrared radiation. Note that there is a large difference between heat and infrared. SEE https://physics.stackexchange.com/questions/80983/how-does-infrared-relate-to-heat.
Here is a link to an excellent explanation of how the absorbed light energy converts/transforms into infrared radiation photons: http://elte.prompt.hu/sites/default/files/tananyagok/InfraredAstronomy/ch03s02.html.
"A molecular vibration is excited when the molecule absorbs a quantum of energy,"
"the vibrational transitions of molecules result in infrared photons [as in absorption filters - added],"
"Molecular transitions result in emission or absorption of photons: the electronic transitions in UV or optical, vibrational transitions in infrared, rotational transitions in microwave range."
Excerpted from:
Toth, Viktor L., Zahorecz, Satolta & Kiss, Csaba. (2013). Infrared Astronomy: Chapter 3.2: The origin of infrared radiation. Hungary: Eötvös Loránd University.
The references from that textbook also contain a huge amount of additional supporting information. Here is a link showing that a simple hydrogen electron can absorb a photon of energy and re-emit a photon of UV, visible light, near IR, mid IR, or far IR. https://chemistry-desk.blogspot.com/2011/05/hydrogen-spectrum.html.
As fluorescent collectors, we know that in UV fluorescence of rocks and minerals, the energy of the UV is absorbed by electrons causing them to oscillate between a high energy state and a lower energy state. The electrons emit visible light photons to shed the energy as they drop from a higher energy state to a lower energy state.
Similarly, in an absorption filter, some of the visible light and some of the ultraviolet energy is absorbed by the electrons and also the molecular bonds causing both of them to oscillate between a higher energy state and a lower state. The electrons and also the molecular bonds emit infrared photons to shed the energy as they drop from a higher energy state to a lower energy state.
There is a very simple demonstration showing that visible light is absorbed by the filter and converted/transformed into infrared radiation - you can find this article titled "A Perspective on Wavelength Transformation by Absorptive Optical Filters." This simple experiment shows that concentrated white light IS converted to IR by the colored glass filter. Please note however, that the IR cameras used for thermal imaging only go down to 8,000 nm, while the range of Infrared radiation starts at 750 nm and goes all the way to 1,000,000 nm. When someone uses an infrared camera to try to show what happens to the energy absorbed by the colored glass filter, they are not measuring any of the wavelengths between 750 nm and 8,000 nm.
Now look at the actual wording in my patent. I claim that the primary band of wavelengths has been transformed and changed while passing through the wavelength transforming material – (i.e. the colored glass filter in this embodiment). I purposely define the UV source to be a UV LED because this patent is specifically limited to UV LEDs. "For the purposes of this invention, it will be understood that the primary UV radiation source is one or more UV light emitting diodes (LED's), and where reference is made to such primary UV radiation source it will be understood to mean an LED source."
Next, I define the wavelength transforming material as an absorbing filter that changes the band of wavelengths and the spectral density. I specifically define one such filter as a band pass filter. The phrase "spectral density" refers to the much lower value of the visible light radiation, the moderately lower value of the UV radiation, and the much higher value of the infrared radiation. "The WT materials (14-14n below) of any preferred embodiments may comprise any material or system that absorbs light of one wavelength or band of wavelengths and emits light of another wavelength or band of wavelengths, thus modifying the distribution of spectral density. Such materials or systems include band pass filters, phosphors and include also such materials or systems such as quantum dots whose properties are a combination of bulk properties and their physical particle size."
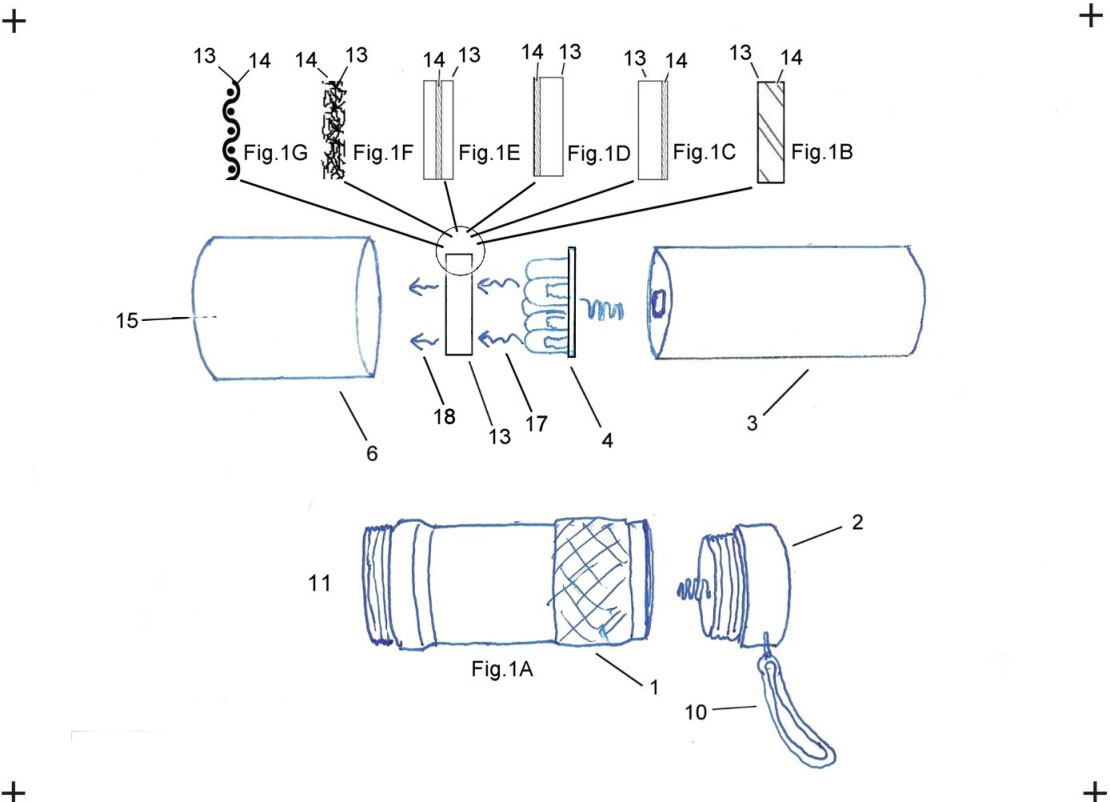
Now consider the LED itself. Examining what the patent actually says, it can be seen that I specifically excluded the dome of the LEDs in my patent with the statement "at least one wavelength-transforming material, deposed external to the envelope of said primary UV LED radiation source, that in response to irradiation by said primary UV LED radiation source, emits a transformed radiation;." Also an examination of the drawings, specifically Figure 1A, shows the LEDs (and their envelopes) as being separate from the wavelength transforming filter. Further, notice that Figure 1A looks remarkably just like the filtered UV LED flashlights that I have been selling since 2006 and it also looks very similar to the Convoy S2+ UV.
Just as you cannot judge a book by its cover, you cannot evaluate a patent by its title. When I was being advised and taught how to write my patents, I was told that the Claims are the heart of the patent. To understand what a patent is teaching, the claims have to be very carefully examined and interpreted. Further, the understanding and interpretation of the Claims has to be modified by the drawings and the written explanation of each feature.
Claim 1
"1. A portable LED apparatus for selectively emitting one or more of a plurality of wavelength distributions of radiation, comprising:
a primary UV LED radiation source, disposed in a housing, wherein said primary UV LED radiation source produces a primary wavelength distribution;
at least one wavelength-transforming material, deposed external to the envelope of said primary UV LED radiation source, that in response to irradiation by said primary UV LED radiation source, emits a transformed radiation;
and wherein said housing further comprises a battery coupled to said primary UV LED radiation source."
I believe that my Claim #1 perfectly describes a UV LED flashlight with a WT colored band pass filter whereby the WT filter absorbs the emission from the primary UV LED source and the WT filter emits a transformed radiation consisting of much less visible light radiation, a little less UV, and much more infrared radiation. Please note that Fig.1B shows a homogenous filter exactly like a colored glass filter wherein the glass is the filter 13, and the colored dye in the band pass filter is the WT material item 14.
The same claim can also refer to other variations wherein the WT filter would emit a transformed radiation with other characteristics. For example: if the filter 13, were homogenously combined with a WT material 14 consisting of a UV reactive phosphor that emits white light, the primary wavelength distribution (a combination of white light radiation, UV radiation, and infrared radiation) would be transformed into much more white light radiation, much less UV radiation, and a little more infrared radiation.
Note also that Fig. 1C & 1D are a representation of a filter which is substrate 13, comprising a glass, plastic, polymer, fluorite, etc. material coupled with a WT material 14, on the surface. If the intent of the filter is to reduce the visible light while transmitting most of the UV, this WT material would be the same or similar to the coating normally seen on BLB lamps/tubes/bulbs. For other embodiments, the WT material can be a UV reactive phosphor to transform the primary band of wavelengths into any number of transformed secondary wavelengths including other wavelengths of UV and also including white light or any color as desired.
This Claim is further strengthened by Figure 1A and by the detailed explanation. FIG. 1A is an exploded perspective view showing a first embodiment of a variable wavelength radiation source, a UV LED Flashlight, apparatus 11, wherein a disk shaped WT filter 13 is coupled with WT materials 14 (as shown in FIGS. 1B-1G) and selection of the radiation to be emitted 18 is made via placing the WT filter 13 between the primary UV radiation source 4 and the exit port of the apparatus 15.
Summary
I firmly believe that my patent is valid and enforceable and that it does properly describe the use of colored glass absorption band pass filters for UV LED flashlights (and similar portable units) for the purpose of reducing the visible light radiation. In addition to that basic embodiment, the patent also goes on to describe other embodiments with WT materials to achieve many other transformations including, but not limited to: shifting from one kind of UV to another kind of UV, or to produce white light, or colored light emissions.
My plan to stop or mitigate the infringement is to 1) educate people that there is a valid patent, 2) to offer filtered UV LED flashlights at a low enough cost so that there is no large profit to be made by infringing, and 3) to offer licenses to people or companies that want to make filtered UV LED flashlights, (or other battery operated filtered UV LED devices).
I want recognition that I do have a patent for the use of WT filters with UV LED flashlights. When people write articles or design DIY webpages that describe putting absorption filters in UV LED Flashlights I want them to also say that this is patented technology.
###
Revised: 2022-10-25